Key Findings at a Glance
Study Objective:
Evaluate sensitivity and clinical performance of deNOVO’s 20X High Sensitive TMB Substrate as an alternative to imported products
Validation Partner:
C-CAMP InDx Centre of Excellence in Quality (Bangalore Baptist Hospital)
Sample Size:
200 well-characterized clinical samples (100 per assay)
Result:
100% concordant performance with imported reference substrate
Recommendation:
“Suitable candidate for substitution over imported TMB”
Study Period:
December 2024 – February 2025
Why is this validation necessary?
For diagnostic manufacturers and in-vitro diagnostics (IVD) developers, substrate quality is non-negotiable.
TMB (3,3′,5,5′-Tetramethylbenzidine) is the chromogenic substrate used in the majority of ELISA-based diagnostic assays worldwide. Its sensitivity, stability, and lot-to-lot consistency directly impact:
- Assay sensitivity (ability to detect low-level analytes)
- Reproducibility (confidence in results across runs)
- Regulatory compliance (validation requirements for CE/FDA approval)
- Clinical accuracy (patient diagnosis and treatment decisions)
Challenges of Indian diagnostic manufacturers
Most high-quality TMB substrates are imported. This creates:
- 6-8 week lead times (vs 1-2 weeks local delivery)
- Import duties and forex volatility (unpredictable pricing)
- Supply chain risks (customs delays, cold chain integrity)
- Lack of technical support (email-only, time zone delays)
The question: Can indigenous TMB substrates match imported quality?
This C-CAMP validation study provides the answer.
The Study:
C-CAMP (Centre for Cellular and Molecular Platforms) is India’s premier life sciences research and innovation hub, supported by the Department of Biotechnology, Government of India.
Their InDx (Indigenisation of Diagnostics) program provides independent validation services for diagnostic reagents and devices.
Why this validation carries weight:
✓ Third-party evaluation (not manufacturer-conducted)
✓ Clinical samples (real patient specimens, not lab standards)
✓ Head-to-head comparison (side-by-side testing with imported reference)
✓ Statistical rigor (Student’s t-test analysis)
✓ Regulatory alignment (validation protocols suitable for regulatory submissions)
Methodology:
Assays Used:
The validation was conducted using two widely adopted commercial ELISA kits:
1. Panbio™ Dengue Early ELISA (Abbott)
Purpose: Detection of NS1 antigen (early infection marker)
Cat. No. 01PE40
2. Panbio™ Dengue IgM Capture ELISA (Abbott)
Purpose: Detection of IgM antibodies (acute infection marker)
Cat. No. 01PE20
Sample Set:
Total samples tested: 200 (100 per assay)
Positive samples: 50 per assay (representing different OD ranges)
Negative samples: 50 per assay
Sample source: Archived clinical specimens from confirmed Dengue patients
(Bangalore Baptist Hospital)
Why Dengue assays?
Dengue diagnostics require high sensitivity – NS1 antigen and IgM antibodies must be detected at low concentrations during critical diagnostic windows. If a TMB substrate performs well in Dengue assays, it will perform well in most diagnostic applications.
Validation Protocol
Step 1: Baseline Testing
Initial testing was performed using serially diluted positive controls to establish baseline performance for both:
-Index substrate (TMB supplied with commercial kit)
-Test substrate (deNOVO 20X High Sensitive TMB)
Step 2: Clinical Sample Testing
Each clinical sample was tested simultaneously using both substrates:
-Same ELISA plate
-Same sample preparation
-Same incubation conditions
-Same reagents (except substrate)
-One set of wells received Index substrate
-One set of wells received Test substrate
Step 3: Data Analysis
Optical density (OD) values were compared using:
-GraphPad Prism (version 10.4.1)
-Student’s t-test for statistical significance
-Scatter plot visualization
Results: 100% Concordance
Diluted Positive Control:
Concordant results observed between test and index substrates across all dilutions.
Clinical Samples:
NS1 Antigen Detection (50 positive, 50 negative):
-100% agreement on positive/negative classification
-No statistically significant difference in OD values (p > 0.05)
=Comparable dynamic range
IgM Antibody Detection (50 positive, 50 negative):
-100% agreement on positive/negative classification
-No statistically significant difference in OD values (p > 0.05)
-Comparable sensitivity across low, medium, and high positive samples
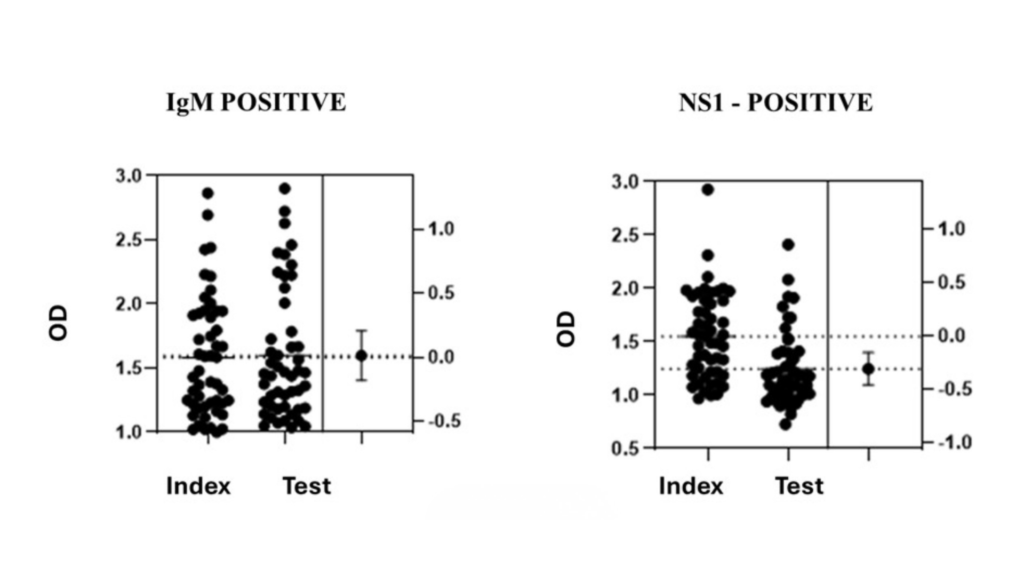
Key Observation:
OD values obtained with deNOVO’s test substrate were statistically equivalent to those obtained with the imported index substrate.
This means:
✓ Same sensitivity for detecting low-level analytes
✓ Same dynamic range for quantitative assays
✓ Same reproducibility across sample types
✓ No false positives or false negatives introduced
C-CAMP’s Conclusion
Official Recommendation:
“Based on the results obtained in this study, we find that 20X High Sensitive TMB would be a suitable candidate for substitution over imported TMB.”
– Dr. Sunil Govekar, CCAMP-InDx CoE Lead
– Dr. Mohammad Nasiruddin, CCAMP-InDx Quality Lead
What it means for Diagnostic Manufacturers?
1. Performance Equivalence
You no longer need to choose between local sourcing and validated performance. deNOVO’s TMB substrate has been independently verified to match imported quality.
2. Supply Chain Advantages
- Delivery: 1-2 weeks (vs 6-8 weeks import)
- Inventory: Local stock availability eliminates stockout risk
- Pricing: No import duties, forex volatility, or freight surcharges
- Support: On-site technical assistance available
3. Regulatory Confidence
This C-CAMP validation provides:
– Independent third-party data for your regulatory submissions
– Clinical sample validation (not just bench testing)
– Statistical analysis suitable for CE/FDA documentation
4. Cost Optimization
Example cost comparison (annual usage for medium-scale IVD manufacturer):
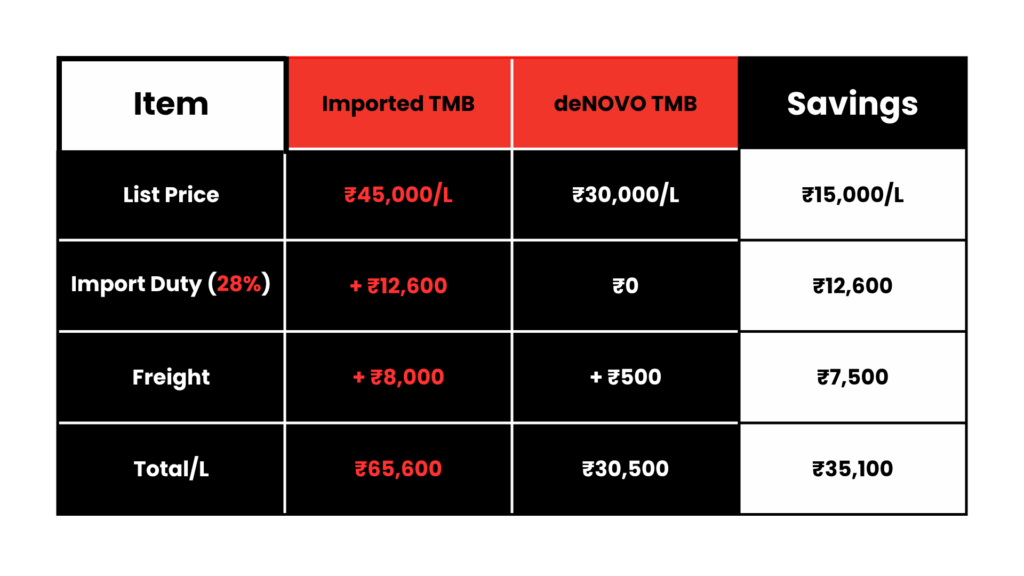
Annual usage (20L): ₹13,12,000 vs ₹6,10,000 = ₹7,02,000 savings
Technical Specifications:
20X High Sensitive TMB Substrate
Concentration:
20X working solution (dilute 1:20 before use)
Formulation:
Optimized for HRP-based ELISA applications
Performance:
-High sensitivity (detects low HRP concentrations)
-Low background (minimal non-specific color development)
-Linear dynamic range (suitable for quantitative assays)
-Stable signal development (consistent color formation kinetics)
Stability:
-Shelf life: 12 months at 2-8°C (unopened)
-Working solution: Stable for 1 month at 2-8°C (after dilution)
Applications:
-Sandwich ELISA
-Competitive ELISA
-Indirect ELISA
-ELISPOT
-Immunohistochemistry (IHC)
-Western blot
Compatible Systems:
-All HRP-conjugated antibodies
-Standard ELISA plate formats (96-well, 384-well)
-Automated and manual systems
Free Sample Evaluation for IVD Developers
We understand the importance of validating reagents in your specific assay systems before switching suppliers.
We offer complimentary sample evaluation:
Request samples: Contact our team with your assay details
Receive evaluation kit: 100mL working solution (sufficient for ~200 assays)
Test in your system: Run side-by-side comparison with your current substrate
Evaluate performance: Assess sensitivity, background, and reproducibility
Get technical support: Our scientists are available for consultation
Request your free evaluation kit:
📞 Phone: +91 80 29575711
📧 Email: info@denovobiolabs.com
🌐 Website: denovobiolabs.com/contact-us
About C-CAMP InDx
The Centre for Cellular and Molecular Platforms (C-CAMP) is an initiative of the Department of Biotechnology, Government of India, established to provide cutting-edge research infrastructure and support for life sciences innovation.
The InDx (Indigenisation of Diagnostics) program focuses on:
-Independent validation of diagnostic reagents and devices
-Supporting indigenous manufacturing through quality assurance
-Building regulatory-ready validation data for Indian diagnostic companies
-Accelerating Make in India initiatives in diagnostics
C-CAMP InDx CoE in Quality operates at Bangalore Baptist Hospital & provides:
-Clinical sample-based validation
-Regulatory-aligned protocols
-Third-party verification services
-Expert guidance from experienced diagnostic scientists
About deNOVO Biolabs
Since 2013, deNOVO Biolabs has been India’s partner in diagnostic and biosimilar innovation.
Our commitment: Prove that indigenous manufacturing can match or exceed global quality standards — not through claims, but through validated data.
Our products:
-ELISA kits (Biosimilar PK, HCP, DNA detection)
-TMB substrates and assay reagents
-Monoclonal and polyclonal antibodies
-Recombinant proteins and antigens
-Custom antibody development services
Our promise:
-Validated performance (independent third-party verification)
-Faster delivery (1-2 weeks vs months of import delays)
-Local support (on-site technical assistance available)
-Competitive pricing (without compromising quality)
Serving:
-6 of India’s top 10 biosimilar companies
-Leading IVD manufacturers
-Government research institutions (DRDO, BARC, DRDE)
-Academic research centers nationwide
Download the Full Validation Report
📄 Download C-CAMP Validation Report (PDF)
Next Steps
For IVD Manufacturers:
1. Request free TMB substrate sample evaluation
2 Download the full C-CAMP validation report
3. Schedule a technical consultation with our team
For Regulatory Teams:
1. Access validation data for your regulatory submissions
2. Discuss vendor qualification requirements
3. Review our quality documentation
For Procurement Teams:
1. Get a detailed cost comparison for your usage volume
2. Discuss inventory management and delivery schedules
3. Explore volume pricing options
Contact us:
📞 +91 80 29575711
📧 info@denovobiolabs.com
📍 A-112, KSSIDC Block-3, Electronics City Phase 1, Bangalore 560100
