
Contents
- How HCP Testing affects Biosimilar Development?
- Generic vs Process-Specific HCP Assays
- Key Selection Criteria for E. Coli HCP ELISA Kits
- Sensitivity Requirements for Regulatory Compliance
- Supply Chain Considerations for Indian Biosimilar Developers
- Validation Framework
- Cost-Benefit Analysis: Import vs Indigenous Manufacturing
- When to Invest in Custom Antibody Development?
How HCP Testing affects Biosimilar Development?
Host cell proteins (HCPs) represent one of the most critical process-related impurities in biopharmaceutical manufacturing.
When you produce recombinant proteins in E. coli expression systems, residual host cell proteins can compromise both product safety and efficacy. According to a 2023 study published in mAbs journal, even trace amounts of HCPsas low as 10-100 parts per million—can trigger immunogenic responses in patients.
The regulatory stakes are high.
The FDA’s guidance on immunogenicity assessment explicitly requires comprehensive HCP monitoring throughout process development and commercial manufacturing. For biosimilar developers filing with CDSCO, EMA, or USFDA, inadequate HCP data can delay approval by months or even years.
Yet choosing the right HCP ELISA kit remains one of the most overlooked decisions in early-stage biosimilar development.
This guide will help you make that decision systematically.
Generic vs Process-Specific HCP Assays
Before you evaluate specific kits, you need to understand the fundamental trade-off in HCP testing.
Generic HCP ELISA Kits
Generic assays use polyclonal antibodies raised against a broad lysate of E. coli proteins. They’re designed to detect a wide variety of host cell proteins regardless of your specific purification process.
Advantages:
- Ready to use immediately (no custom development timeline)
- Validated across multiple E. coli strains
- Cost-effective for process development
- Regulatory precedent (widely accepted when properly validated)
Limitations:
- May not detect all process-specific HCPs
- Antibody coverage varies by kit manufacturer
- Requires validation for your specific matrix
Process-Specific HCP Assays
Process-specific assays are developed using HCPs isolated directly from your purification process. The antibodies are raised against your actual impurity profile.
Advantages:
- Maximum coverage of process-relevant HCPs
- Tailored to your specific purification conditions
- Highest sensitivity for critical low-abundance impurities
Limitations:
- 6-12 month development timeline
- Significantly higher cost ($50,000-$150,000 USD)
- Risk of missing unanticipated impurities that emerge from process changes
According to FDA guidance on HCP assays, both approaches are acceptable, provided you demonstrate adequate coverage through orthogonal methods like mass spectrometry or 2D Western blot.
Key Selection Criteria for E. Coli HCP ELISA Kits
When evaluating HCP ELISA kits, five factors drive your decision.
1. Sensitivity & Detection Range
Regulatory agencies typically require HCP levels below 100 ppm (parts per million) in final drug substance. For a 10 mg/mL biologics product, that translates to <1,000 ng/mL HCP concentration.
Your ELISA kit must reliably quantify at or below this threshold.
What to look for:
- Lower limit of quantitation (LLOQ) ≤ 5 ng/mL
- Detection range spanning 3-4 orders of magnitude
- Validated precision (CV <20%) across the range
Industry benchmark: Most established kits offer detection ranges of 3.9-500 ng/mL or 7.8-500 ng/mL. DeQuanto® E. Coli HCP ELISA Kit achieves 1.95-500 ng/mL providing additional sensitivity margin for highly purified samples.
2. Validated E. Coli Strain Coverage
E. coli strains vary in their protein expression profiles. Your HCP kit must recognize proteins from YOUR specific expression strain.
Common biosimilar expression strains:
- BL21(DE3) and derivatives
- DH5α
- K12
- JM109
- TOP10F
- MC1061
According to research published in Biotechnology and Bioengineering, HCP profiles can vary by 15-25% between strains, even within the same species.
Best practice: Confirm that your kit manufacturer has validated antibody reactivity against your specific strain using Western blot or orthogonal methods.
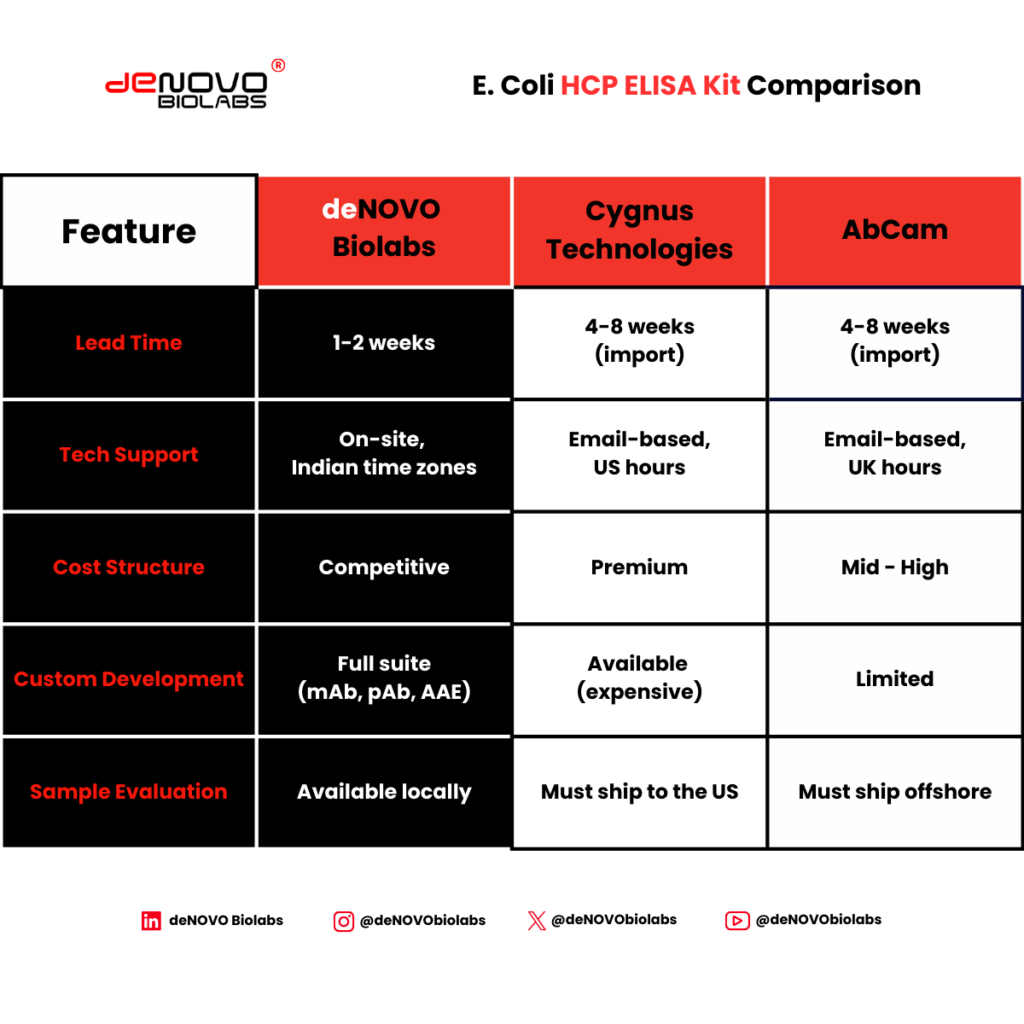
3. Supply Chain Reliability
This factor is often ignored—until it becomes critical.
A 2024 survey by BioPlan Associates found that 38% of biosimilar developers experienced project delays due to reagent supply interruptions. The average delay? 6.2 weeks.
Questions to ask:
- What is the typical lead time from order to delivery?
- Does the supplier maintain safety stock?
- What happens if I run out mid-batch?
- Are there import/export restrictions?
- What are the cold chain requirements?
For Indian biosimilar developers, importing kits from the US or EU typically involves:
- 4-8 week lead times
- Cold chain logistics (2-8°C maintenance)
- Customs clearance variability
- Forex exposure on pricing
Indigenous alternative: deNOVO Biolabs manufactures DeQuanto® HCP kits in Bangalore with 1-2 week delivery across India—eliminating import delays and cold chain risks.
4. Technical Support During Validation
Every HCP assay, regardless of manufacturer, must be validated for YOUR specific sample matrix per ICH Q6B guidelines.
Critical validation studies include:
- Dilutional linearity testing
- Matrix interference assessment
- Spike-and-recovery experiments
- Precision and accuracy determination
- Antibody coverage evaluation
The question is: Will your supplier help you execute these studies?
Email-based support from overseas vendors often means:
- 12-24 hour response times (time zone differences)
- Generic troubleshooting templates
- No on-site assistance for method transfer
Local advantage: deNOVO provides on-site technical support for assay qualification – our scientists can visit your facility in Hyderabad, Ahmedabad, Pune, or other major biotech hubs to assist with validation protocols.
5. Total Cost of Ownership
List price is only one component of total cost.
Hidden costs to consider:
- Import duties and taxes (28% GST + customs duty for lab reagents in India)
- Freight and cold chain logistics
- Inventory holding costs (if you overstock to avoid stockouts)
- Opportunity cost of delayed timelines
- Validation study costs
Example calculation for a typical process development program:
| Cost Component | Imported Kit | Indigenous Kit (deNOVO) |
|---|---|---|
| List price per kit | ₹1,80,000 | ₹1,20,000 |
| Import duties (28%) | ₹50,400 | ₹0 |
| Freight & cold chain | ₹15,000 | ₹2,000 |
| Total per kit | ₹2,45,400 | ₹1,22,000 |
| Lead time | 6-8 weeks | 1-2 weeks |
| Technical support | Email only | On-site available |
Over a 12-month process development program using 10 kits, the cost difference is ₹12.34 lakh—enough to fund an additional purification campaign.
Sensitivity Requirements for Regulatory Compliance
Regulatory agencies don’t specify exact HCP limits, but they expect you to justify yours based on product dosing and safety data.
Typical HCP Acceptance Criteria
For parenteral biologics:
- Final drug substance: <100 ppm (ng HCP per mg product protein)
- Intermediate purification steps: <1,000-10,000 ppm
For high-dose products (>10 mg/kg):
- Even stricter limits may be required (<10-50 ppm)
According to EMA guidelines on quality of biological medicinal products, you must demonstrate that your analytical method can detect HCPs at levels significantly below your acceptance criteria.
The 10X rule: If your acceptance criterion is 100 ppm, your assay should quantify reliably at ≤10 ppm.
For a 10 mg/mL product, that means detecting <100 ng/mL HCP.
This is where kit sensitivity becomes critical. A kit with LLOQ of 7.8 ng/mL gives you ~10-fold margin. A kit with LLOQ of 1.95 ng/mL gives you ~50-fold margin—providing greater confidence at late purification stages.
Supply Chain Considerations for Indian Biosimilar Developers
India’s biosimilar industry is growing rapidly. According to IBEF (India Brand Equity Foundation), India’s biosimilar market is projected to reach $30 billion by 2030, growing at 22% CAGR.
Yet most Indian biosimilar developers still import 70-80% of their critical reagents.
The supply chain vulnerabilities are real:
- Geopolitical disruptions – COVID-19 demonstrated how quickly global supply chains can freeze
- Regulatory changes – Import restrictions on certain biologics reagents
- Forex volatility – INR depreciation increases costs unpredictably
- Port congestion – Bangalore-based developers wait for shipments through Chennai or Mumbai ports
- Cold chain failures – Temperature excursions during transit render kits unusable
The Make in India opportunity:
Indigenous manufacturing eliminates these vulnerabilities. deNOVO Biolabs was founded in 2013 as one of India’s first biotech startups under the BIRAC initiative, specifically to address this gap.
Today, 6 of India’s top 10 biosimilar companies use deNOVO’s ELISA kits and custom antibody services.
Key advantages of local sourcing:
- 1-2 week delivery (vs 6-8 weeks import)
- No cold chain risk (direct truck delivery, 24-48 hours to tier-1 cities)
- Rupee-denominated pricing (no forex exposure)
- On-site technical support (validation assistance at your facility)
- Sample evaluation (try before you commit—we’ll run your samples)
Validation Framework
Regardless of which kit you choose, validation is non-negotiable.
ICH Q2(R1) Analytical Validation requires you to demonstrate:
1. Dilutional Linearity
Test: Dilute a high-HCP sample serially (1:10, 1:100, 1:1000) and measure each dilution.
Acceptance: Calculated HCP concentration should be within ±20% across dilutions.
Why it matters: Non-linear dilution indicates matrix interference or antibody saturation.
2. Matrix Recovery (Spike-and-Recovery)
Test: Spike a known amount of HCP standard into your actual sample matrix (your purification buffer with your product).
Acceptance: Recovery should be 80-120%.
Why it matters: Low recovery means your matrix interferes with antibody binding. High recovery suggests non-specific binding.
Common interfering substances:
- High salt concentrations (>500 mM NaCl)
- Detergents (Tween-20, Triton X-100)
- Urea or guanidine (from refolding buffers)
- Extreme pH (<5.0 or >8.5)
- Organic solvents
If you see poor recovery, you may need to:
- Dilute samples further
- Adjust buffer composition
- Use a different diluent
- Consider custom antibody development
3. Precision Studies
Test: Run the same sample 6-10 times in the same run (intra-assay) and across 3 different runs (inter-assay).
Acceptance: Coefficient of variation (CV) <20% for concentrations above LLOQ.
Why it matters: High variability compromises your ability to detect HCP reduction during process optimization.
4. Antibody Coverage Assessment
This is the most important and most neglected validation study.
Generic HCP kits can’t detect every HCP. The question is: Do they detect the HCPs that matter in YOUR process?
Methods to assess coverage:
Option A: Antibody Affinity Extraction (AAE)
- Incubate HCP sample with kit antibodies
- Remove antibody-bound HCPs
- Analyze remaining (unbound) HCPs by mass spectrometry or SDS-PAGE
- Calculate % depletion
Target: >70-80% depletion indicates good coverage
Option B: 2D Western Blot
- Run HCP sample on 2D gel (separates by charge and size)
- Probe with kit antibodies
- Compare to silver stain (shows ALL proteins)
- Count spots: antibody-detected vs total
Target: >70% of visible HCP spots should be antibody-reactive
deNOVO offers AAE services to help you qualify generic HCP kits for your specific process ensuring you’re not missing critical impurities.
When to invest in Custom Antibody Development?
Generic HCP kits work for most standard biosimilar processes. But sometimes, you need custom antibodies.
Consider custom development if:
Red Flag #1: Poor Matrix Recovery (<60%)
If your spike-recovery study shows <60% recovery even after optimization, your sample matrix is fundamentally incompatible with the generic kit antibodies.
Red Flag #2: Low Antibody Coverage (<50%)
If AAE or 2D Western blot shows the kit antibodies only recognize <50% of your process HCPs, you’re missing critical impurities.
Red Flag #3: Regulatory Pushback
If FDA, EMA, or CDSCO reviewers question your HCP coverage during IND/IMPD/CTD review, you may need process-specific data.
Red Flag #4: Novel Purification Process
If you’re using unconventional methods (novel affinity ligands, extreme pH, high organic solvents), your HCP profile may be atypical.
Custom antibody development timeline:
- Immunization: 8-12 weeks
- Antibody screening & validation: 4-6 weeks
- Assay development & qualification: 6-8 weeks
- Total: 18-26 weeks
deNOVO offers full custom antibody services including:
- Polyclonal antibody development (rabbit, goat, chicken)
- Monoclonal antibody development (mouse, rat, humanized)
- Hybridoma development and banking
- ELISA assay development and validation
- GMP-scale antibody production
Contact us for a technical consultation: info@denovobiolabs.com
The DeQuanto® E. Coli HCP ELISA Kit Advantage
At deNOVO Biolabs, we understand the challenges biosimilar developers face because we’ve been there.
Founded in 2013 as part of Karnataka’s pioneering biotech startup ecosystem, we became India’s first company to manufacture ELISA kits for biosimilar pharmacokinetics and immunogenicity testing.
Today, we serve 6 of India’s top 10 biosimilar companies with products and services that match—or exceed—global standards.
DeQuanto® E. Coli HCP ELISA Kit Specifications
Sensitivity: 1.95 – 500 ng/mL detection range
Validated Strains: BL21, DH5α, JM109, TOP10F, K12, MC1061
Precision: CV <15% (intra-assay), CV <20% (inter-assay)
Hook Effect Threshold: >200 µg/mL (higher than most competitors)
Delivery: 1-2 weeks across India
Format: 96-well strip plates (flexible usage)
Regulatory: Suitable for process development and method validation for CDSCO/EMA/FDA filings
View full product specifications →
Why Biosimilar Developers choose us?
Speed: When your batch is ready and you’re out of kits, 1-2 week delivery beats 6-8 week import timelines.
Support: Our scientists provide on-site assay qualification support—we’ll come to your lab in Bangalore, Hyderabad, Ahmedabad, or Pune.
Flexibility: Need custom antibody development? We integrate that seamlessly. Want to validate our kit against your current supplier? We’ll run side-by-side studies.
Local: No import duties. No cold chain risk. No forex exposure. No time zone delays for technical support.
Quality: ISO-certified manufacturing. Validated across multiple biosimilar processes. Proven performance in regulatory filings.
Beyond HCP Kits
While HCP testing is critical, it’s just one piece of your analytical toolkit.
deNOVO provides a complete suite of biosimilar development services:
Antibody Development Services
- Custom monoclonal & polyclonal antibody development
- Hybridoma development & subcloning
- Antibody characterization (epitope mapping, affinity determination)
- Large-scale antibody production (mg to gram quantities)
Learn more about antibody services →
Cell-Based Assays
- Neutralizing antibody assays (NAb)
- Potency assays for biosimilars
- Antibody-dependent cell-mediated cytotoxicity (ADCC)
- Complement-dependent cytotoxicity (CDC)
Explore cell-based assay capabilities →
Lateral Flow Assay Development
- Rapid test development for biosimilar monitoring
- Custom LFA for in-process QC
- Gold nanoparticle conjugation
- Strip manufacturing & validation
View LFA development services →
Recombinant Protein Production
- E. coli expression systems
- Mammalian cell expression
- Protein purification & characterization
- Endotoxin removal to <0.1 EU/mg
See recombinant protein catalog →
Ready to Optimize Your HCP Testing Strategy?
Choosing the right HCP ELISA kit is about building a partnership that accelerates your biosimilar development timeline while meeting regulatory requirements.
Whether you need:
- A validated generic HCP kit for immediate use
- Technical support for assay qualification
- Custom antibody development for process-specific needs
- A second opinion on your current HCP testing strategy
Our team is here to help.
📞 Schedule a Technical Consultation
Email: info@denovobiolabs.com
Phone: +91 80 29575711
Address: A-112, KSSIDC Block-3, Electronics City Phase 1, Bangalore 560100
Business Hours: Monday-Friday, 9:00 AM – 6:00 PM IST
About deNOVO Biolabs
Since 2013, deNOVO Biolabs has been at the forefront of India’s biosimilar revolution.
As one of the earliest biotech startups supported by BIRAC and Karnataka’s Vision Group, we pioneered indigenous manufacturing of critical biosimilar reagents—making India less dependent on imports and accelerating local drug development.
Today, we serve:
- 6 of India’s top 10 biosimilar companies
- Premier government research institutions (DRDE, DRDO, BARC)
- Academic research centers across India
- Diagnostic manufacturers in emerging markets
Our mission: Make healthcare accessible by enabling faster, more affordable biosimilar development through world-class reagents and services manufactured in India.
Discover. Innovate. Collaborate.
